24
2014-11
BrosMed Announces the Launch of a New Company Logo and Website
November 24, 2014, we are proud to announce the launch of a new company logo and release our new website, designed with a fresh new look and user-friendly navigation, updated with the latest information about our products and services, as part of the ongoing evolution of our company’s brand and Visual Identity (VI).
2014-11-24
08
2014-10
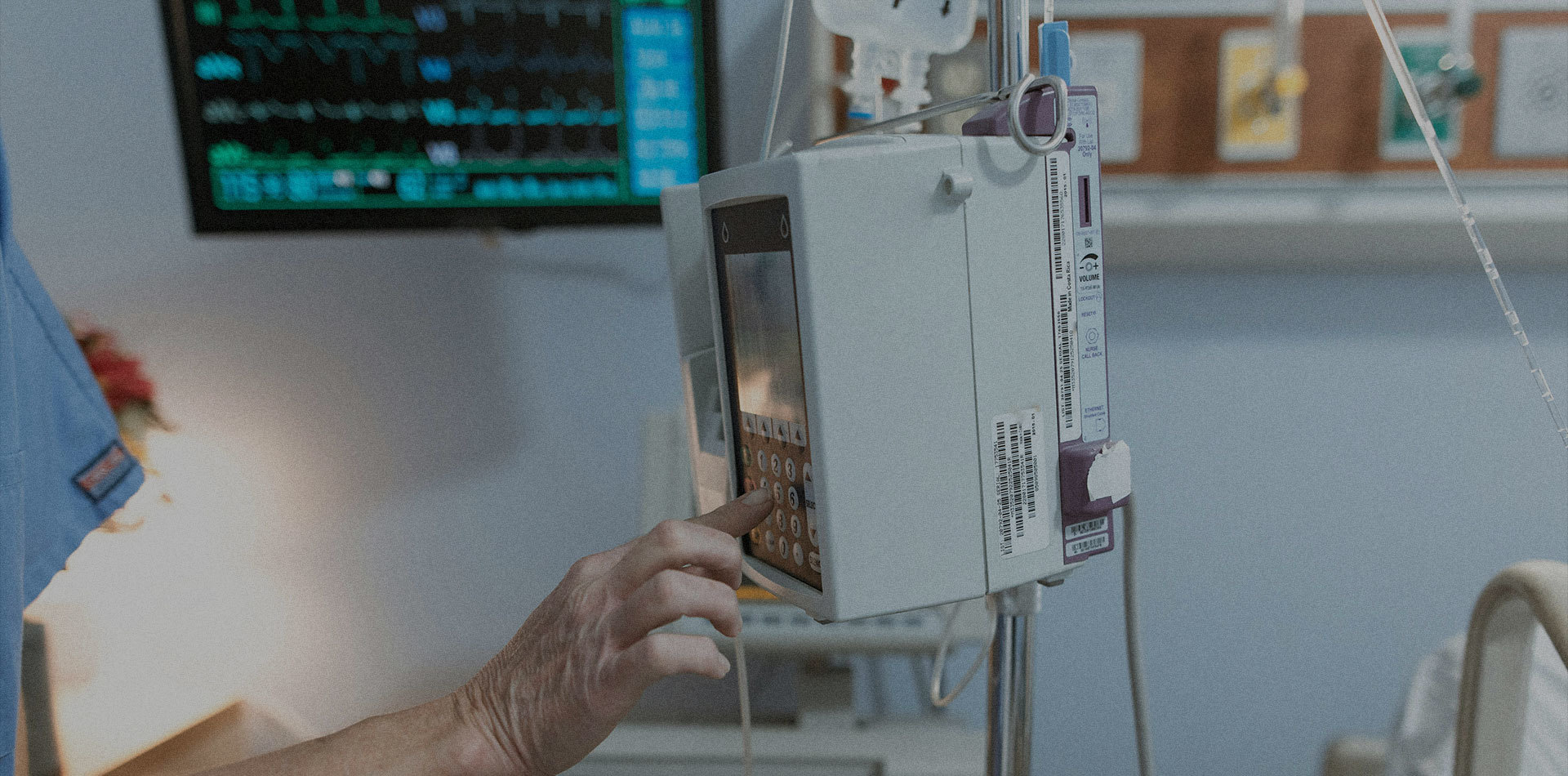
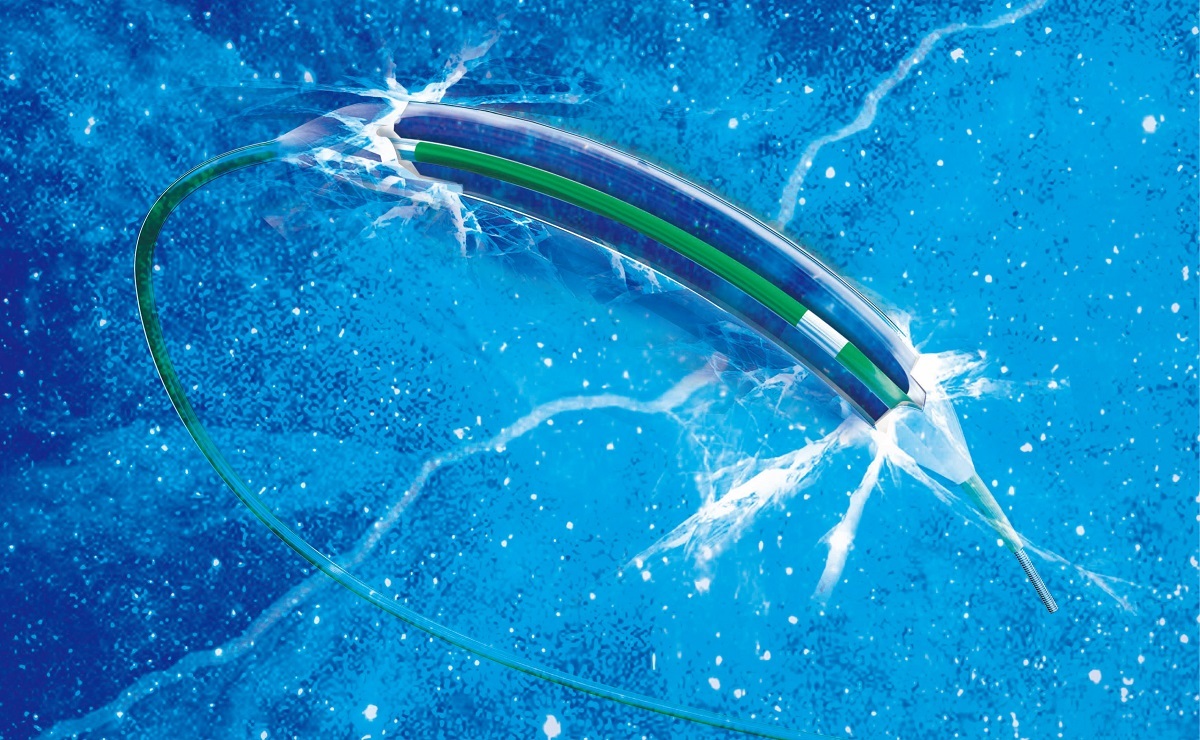
October 8, 2014— Following the FDA approval of Artimes™ Balloon Dilatation Catheter in September, BrosMed announced FDA 510(k) clearance for Apollo™ Balloon Dilatation Catheter which is now approved and ready for sale in the United States of America and the North America market for the interventional cardiovascular use.
2014-10-08
17
2014-09
September 17, 2014— Following the CE approval and launch Artimes™ Balloon Dilatation Catheter, BrosMed announced FDA 510(k) clearance for Artimes™ Balloon Dilatation Catheter which is now approved and ready for sale in the United States of America and the North America market for the interventional cardiovascular use.
2014-09-17
02
2014-09
BrosMed Establishes Europe Subsidiary in the Netherlands
September 2, 2014, BrosMed Medical has announced the establishment of a subsidiary company in the Netherlands, BrosMed Medical B.V.
2014-09-02
10
2014-07
July, 10, 2014— Following the CE approval and launch Artimes™ and Apollo™ Balloon Dilatation Catheter, BrosMed announced CE Mark and Launch of Fourteen (14) types of interventional accessories which are now approved in EU for the cardiology and radiology procedures.
2014-07-10
25
2014-02
BrosMed Announces CE Mark and Launch for Apollo™ Balloon Dilatation Catheter
February 25, 2014— Following the CE approval and launch Artimes™ Balloon Dilatation Catheter, BrosMed announced CE Mark and Launch of its Apollo™ Balloon Dilatation Catheter which is now approved in the EU for the interventional cardiovascular use.
2014-02-25
21
2013-10
BrosMed Announces CE Mark and Launch for Artimes™ Balloon Dilatation Catheter
October 19, 2013– BrosMed announced CE Mark and launch of its Artimes™ Balloon Dilatation Catheter, which is now approved in the EU for the of improving myocardial blood flow in the localized stenotic lesion of the coronary arteries.
2013-10-21
16
2013-07
BrosMed Received FDA Initial Registration for Facility Establishment
July 16, 2013, BrosMed announced that the company received registration notification identifying BrosMed Medical facility establishment by the U.S. Food and Drug Administration (FDA) Center for Devices and Radiological Health (CDRH). The owner operator number is 10044695. Currently company is pursuing FDA approval for a series of products in order to enter North America markets.
2013-07-16